Published On Aug 3, 2017
MIT 5.111 Principles of Chemical Science, Fall 2014
View the complete course: https://ocw.mit.edu/5-111F14
Instructor: Catherine Drennan
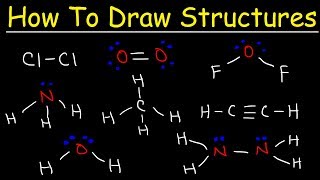
Lewis structures are simplistic views of molecular structure. They are based on the idea that the key to covalent bonding is electron sharing and having each atom achieve a noble gas electron configuration. Lewis structures correctly predict electron configurations around atoms in molecules about 90% of the time. They are not perfect, but writing a Lewis structure is a lot easier than solving the Schrödinger equation, so we recommend that you watch this lecture.
License: Creative Commons BY-NC-SA
More information at http://ocw.mit.edu/terms
More courses at http://ocw.mit.edu